 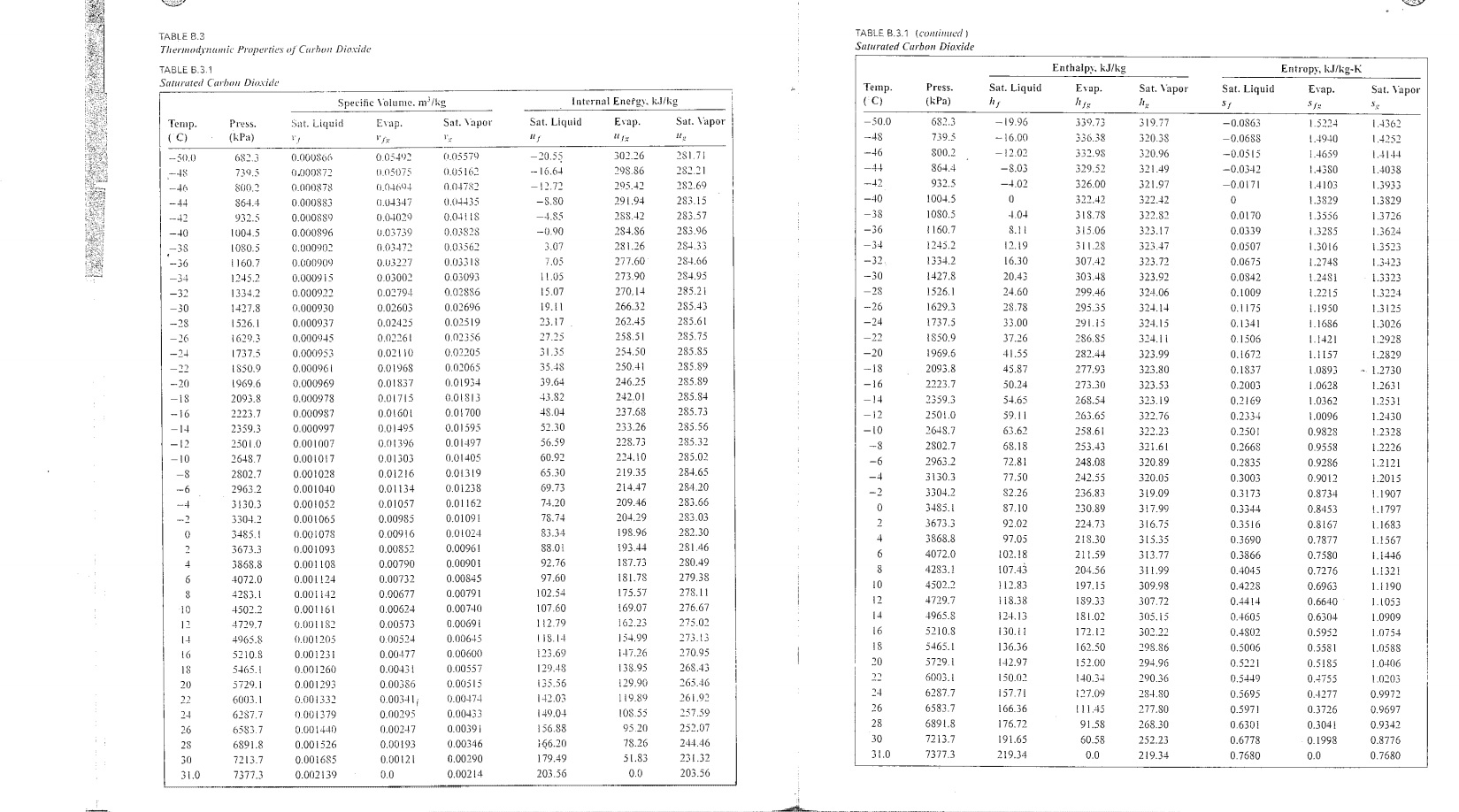
T Ps C, Ifg I sf sg h s - I [kJ/kg K1 [kJ/kgl [kJ!kg K l 
per unit amount-of-substance) 8 -refers to a property at standard pressure pe = 1 bar (the superscript o is often used) velocity of sound -specific, molar heat capacity at constant p -specific, molar heat capacity at constant u - specific, molar Gibbs function (h - Ts, g- T?) -molar Gibbs function of reaction, of formation - specific, molar enthalpy (u + pu, ii + pi?) - molar enthalpy of reaction, of formation - equilibrium constant, of formation - thermal conductivity - molar mass -absolute pressure - Prandtl number (cpp/k) - specific, molar (universal) gas constant -specific, molar entropy -absolute temperature (K) or Celsius temperature ("C) -temperature interval or difference - specific, molar internal energy -specific, molar volume (lip, 1/p) -geometric altitude above sea level -ratio of specific heat capacities (cp/c, = Ep/E,) -mean free path -dynamic viscosity -kinematic viscosity (PIP) - mass, molar density (1 /v, 116)Ĭ -refers to a property in the critical state f -refers to a property of the saturated liquid, or to a value of formation g -refers to a property of the saturated vapour fg -refers to a change of phase at constant p I -refers to a property of the saturated solid s -refers to a saturation temperature or pressure KJ/kg K, kJ/kmol K kJ/kg K, kJ/kmol K K or "C K kJ/kg, kJ/kmol m3/kg, m3/kmol m 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
